Identify The Bronsted Lowry Acid In The Following Reaction
We are also able to handle any complex paper in any course as we have employed professional writers who are specialized in different fields of study. Identify the Brønsted acid Brønsted base conjugate acid and conjugate base in an acid-base reaction.

Lipid Metabolism Lipid Metabolism Biochemistry Chemistry
Identify the conjugate acid-base pairs in the following reactions.

Identify the bronsted lowry acid in the following reaction. A Bronsted-Lowry acid is a substance that gives away protons hydrogen cations textH and is therefore called a proton donor. Acid and base virtual lab answers. Acid strength is determined by the amount of that acid that actually ionizes.
HCl HI HBr HNO_3 H_2SO_4 and HClO_4. H OH- H 2 O Hydroxide donates the pair of electrons - Lewis base Hydrogen ion accepts the pair of electrons Lewis acid Identify the Lewis acid and Lewis Base in the following reaction. Water reacts with a base to form the hydroxide ion.
The Brønsted-Lowry theory is defined by the following reaction. The products of the reaction are a salt and water. An acid and a base form water and salt.
The conjugate base that we made in the reaction above HSO 4 can dissociate further and be an acid in a different reaction. The Bronsted-Lowry model defines acids and bases in terms of their ability to donate or accept protons. Phenolphthalein is the indicator used in acid-base titration in order to identify whether solution is acid or base.
Do not be fooled by thinking that H indicates acid and OH indicates base though some common inorganic compounds are often written this way. We offer assignment help in more than 80 courses. Ask questions to evaluate merits and limitations of the Arrhenius and Bronsted-Lowry models of acid and bases.
Answer 1 of 5. Acid base i H 2O NH 3 OH- NH 4. HCO3- H3PO4 aq H2CO3 aq H2PO4- The formula for the conjugate of HCO3- is.
Which of the following is true about the results of a neutralization reaction. Preview 15 - pH Value in case of Strong Acid Strong Base. First consider the simplified general equation of a simple acid reaction.
Acidity Here are some general guidelines of principles to look for that can help you address the issue of acidity. Preview 16 - pH value of a Mixed Solution. Definition Example 309 Lewis Acid.
According to Newtons third law for every action force there is an equal in size and opposite in direction reaction forceForces always come in pairs - known as action-reaction force pairs Identifying and describing action-reaction force pairs is a simple matter of identifying the two interacting objects and making two statements describing who is pushing on whom and in what direction. Acid and base virtual lab answers. Preview 12 - Concept of Acid Base.
A redox reaction is a chemical reaction that involves a transfer of electrons and changes in oxidation number. Learn about redox reactions and identify. A conjugate base forms after the acid loses a proton while the conjugate acid forms when the base.
Definition Examples 616 Exothermic Reaction. Chapter 14 review acids and bases answer key. Momentum as a Vector Quantity.
Draw a lewis dot structure for. Key Takeaways Key Points. Acid and base virtual lab answers.
The products of the reaction are a salt and water. You can choose your academic level. Look at this equation and see how it fits the Bronsted-Lowry and Lewis definitions.
Acids and bases interact with each other in what is called a neutralization reaction. There are 6 that most consider to be the STRONG acids. 91 KCl 92 MgNO 3 2 93 CaSO 4.
Preview 14 - pH value and Effect of Temperature on pH value. Here the indicator shows colourless to pink indicates that the medium is basic and pH is 10 which is greater than 7. To fully describe the momentum of a 5-kg bowling ball moving westward at 2 ms you must include information about both the magnitude and the direction of the bowling ball.
From their experience they are able to work on the most difficult assignments. High school collegeuniversity masters or pHD and we will assign you a writer who can satisfactorily meet your professors expectations. For example HCl.
Plan and carry out an investigation to explore acid-base neutralization. The following are some of the course we offer assignment help in. Sort of but only if you actually know some chemistry and have the structural formula as opposed to the molecular formula.
In the following net ionic equation identify each species as either a Brønsted-Lowry acid or a Brønsted-Lowry base. The Ksp for cerium iodate CeIO33 is 32 e-10. Option c is the answer.
Bronsted-Lowry acid proton donor H proton Bronsted-Lowry base proton acceptor H proton Bronsted-Lowry acid-base reaction reaction involving the transfer of a proton TASK 1 Bronsted-Lowry acids bases Identify the Bronsted-Lowry acid and base in each of the following reactions. 11 - Concept of Acid Base. All Bronsted-Lowry acids and bases are also Lewis acids and bases but not necessarily the other way around.
Bronsted-Lowry and Lewis theories of acids and bases and explains the. Preview 13 - Ostwalds Dilution Law. In each case give an acid-base reaction equation that could be used to produce.
Acids are molecular covalent compounds which you dont expect to ionize release an H and leave behind the conjugate base or Cl- for example. Thus once an acid loses the proton it becomes a conjugate baseAnd once a base accepts a proton it becomes a conjugate acidRemember that when we are talking about the conjugates we are always talking about the products of a specific acid-base reaction. We always make sure that writers follow all your instructions precisely.
Definition Theory Examples 459. Predict the major product of the following reaction h2o h2so4. Momentum is a vector quantityAs discussed in an earlier unit a vector quantity is a quantity that is fully described by both magnitude and direction.
The pH is neutralized to 7 if both the acid and base fully react. Water reacts with an acid to form the hydronium. The strongest acids ionize 100.
Conceptual understanding of pH as related to acidbasic conditions is needed g. Labster advanced acids and bases. Acid base conjugate base conjugate acid.
31 -CH 3 COO- H 2.

Lipid Metabolism Lipid Metabolism Biochemistry Chemistry

E1 Reaction Of Cyclohexanes Produces Zaitsev Product Chemistry Methyl Group Chemistry Notes

Two Chemical Equations Demonstrating Proton Transfer In Water In The First Equation Water Autoi Middle School Chemistry Chemistry Classroom Chemical Changes

Diagramming Galvanic Cells Handout And Worksheet Cells Worksheet Biology Worksheet Chemistry Worksheets

Naming Bridged Bicyclic Compounds Master Organic Chemistry Organic Chemistry Chemistry Organic Chemistry Study

Teaching Chemistry Chemistry Worksheets Science Teaching Resources

The Prefix Cyclo Is Combined With The Suffix Ene Notice That The Numbering Starts From The Double Bond And Goes Study Chemistry Chemistry Lessons Chemistry

Bulky And Non Bulky Bases In Sn1 Sn2 E1 And E2 Mechanism How To Memorize Things Sn1 Sn2 Chemistry

Chemthink Lockscreen Chemistry Projects To Try

Le Chatelier S Principle Animation Here Le Chatelier S Principle Teaching Chemistry Chemistry

Organic Chemistry Functional Groups Cheat Sheet Organic Chemistry Organic Chemistry Study Teaching Chemistry

Pin On Nmr Spectroscopy Practice Problems

List Of Chemical Formula Or Common Name Of All Inorganic Compound Brainly In Chemistry Basics Science Formulas Chemistry Lessons

Chemistry Now Chiral Molecules Chemistry Science Education Study Materials

Difference Between Sn1 And Sn2 Reactions Chemistry Notes Organic Chemistry Reactions

Examples Morphology Of Flowering Plants Part 2 Biology Notes Neet Notes Study Biology

Sample Resume Resume Examples Resume

Nuclear Stability And Magic Numbers Physical Chemistry Chemistry Nuclear

Difference Between Sn1 And Sn2 Reactions Chemistry Notes Organic Chemistry Reactions

R S Configuration Absolute Form R S Represention Enantiomer Diatereomer Non Superimposable Non Mirror Image Chemistry Notes Chemistry Organic Chemistry

Pin By Dinkx On Price School Persuasive Writing Prompts Teaching Chemistry Physical Science Middle School

Methane Burning Methane Math Fundamental

How To Identify Different Types Of Chemical Compounds Youtube Science Notes Chemistry Class Chemistry

When Two Immiscible Liquids Are Placed In A Separatory Funnel Two Layers Are Seen The Denser Solvent Will Be Solutions And Mixtures Chemistry Chemistry Labs

One Pager How To Use One Pagers In The Science Classroom This Project Is Perfect For Your Kids T Middle School Science Classroom Science Classroom One Pager
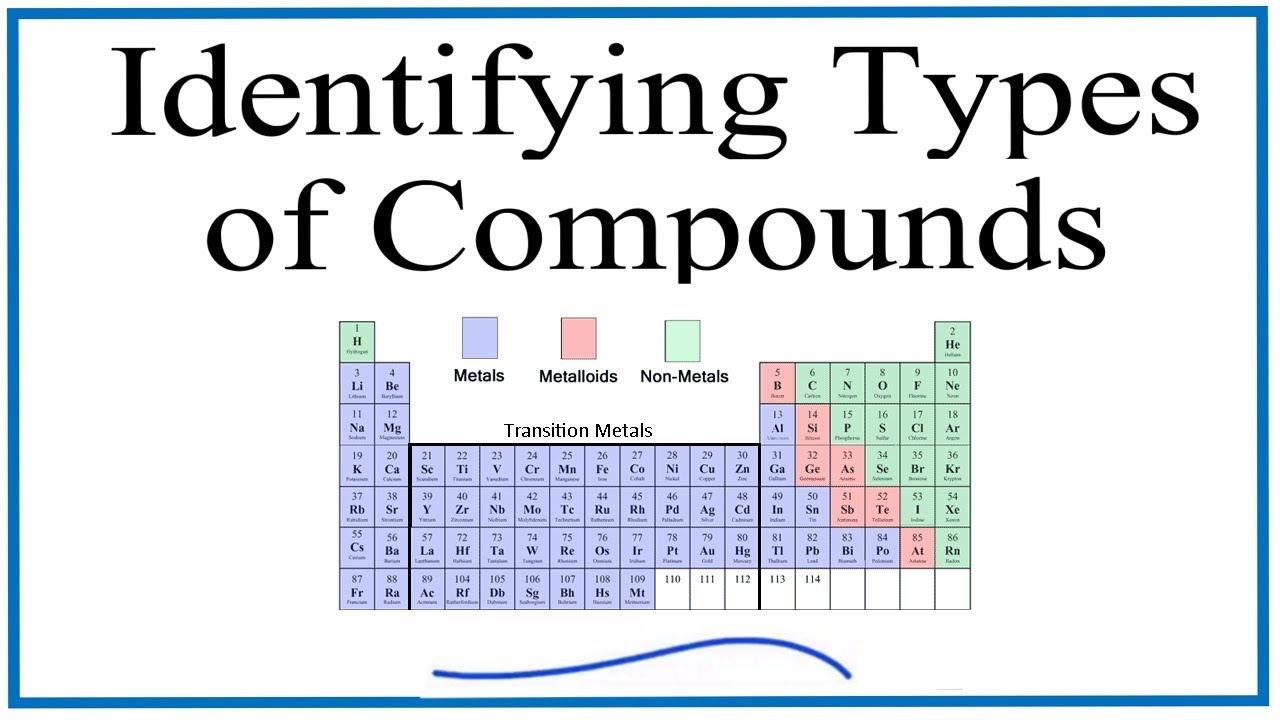
How To Identify Different Types Of Chemical Compounds Youtube Science Notes Chemistry Class Chemistry

How To Identify Different Types Of Chemical Compounds Youtube Science Notes Chemistry Class Chemistry

The Prefix Cyclo Is Combined With The Suffix Ene Notice That The Numbering Starts From The Double Bond And Goes Study Chemistry Chemistry Lessons Chemistry

Definitions Of Arrhenius Bronsted Lowry And Lewis Acids And Bases In Organic Chemistry Organic Chemistry Organic Chemistry Study Teaching Chemistry

Amino Acids Chemistry Biochemistry Nutrition Biochemistry Chemistry Protein Biology

Bbc Bitesize Ks3 Chemistry The Rock Cycle Revision 5 Rock Cycle Physical Weathering Weathering And Erosion

The Prefix Cyclo Is Combined With The Suffix Ene Notice That The Numbering Starts From The Double Bond And Goes Study Chemistry Chemistry Lessons Chemistry

The Prefix Cyclo Is Combined With The Suffix Ene Notice That The Numbering Starts From The Double Bond And Goes Study Chemistry Chemistry Lessons Chemistry

Chemia Energia Pracovny List Sacharidy Otazky A Ulohy List Inbox Screenshot

Aldrin Is An Organochlorine Insecticide That Was Widely Used Until The 1990s When It Was Banned In Most Countries It Is A Colourless Solid Before T Chemie

Pin On Acids And Bases In Organic Chemistry

The Prefix Cyclo Is Combined With The Suffix Ene Notice That The Numbering Starts From The Double Bond And Goes Study Chemistry Chemistry Lessons Chemistry

Examples Morphology Of Flowering Plants Part 2 Biology Notes Neet Notes Study Biology

Highschool Chemie Kernkonzept Spickzettel 15 Chemische Bindung Bindung Chemie Chemische Chemistry Worksheets Teaching Chemistry Chemistry Education

The Prefix Cyclo Is Combined With The Suffix Ene Notice That The Numbering Starts From The Double Bond And Goes Study Chemistry Chemistry Lessons Chemistry

What Do Ortho Meta And Para Mean In Organic Chemistry Organic Chemistry Study Chemistry Paper Chemistry Notes

Acids And Bases Teaching Chemistry Chemistry Education Chemistry Lessons

Ir Spectroscopy Functional Groups Ir Absorbance Table Http Www Chromatographytechniques Com Articles Functional Group Chemistry Help Mcat Study

Pin By Lizi Glurjidze On My Saves Ideal Gas Law Kinetic Theory Root Mean Square
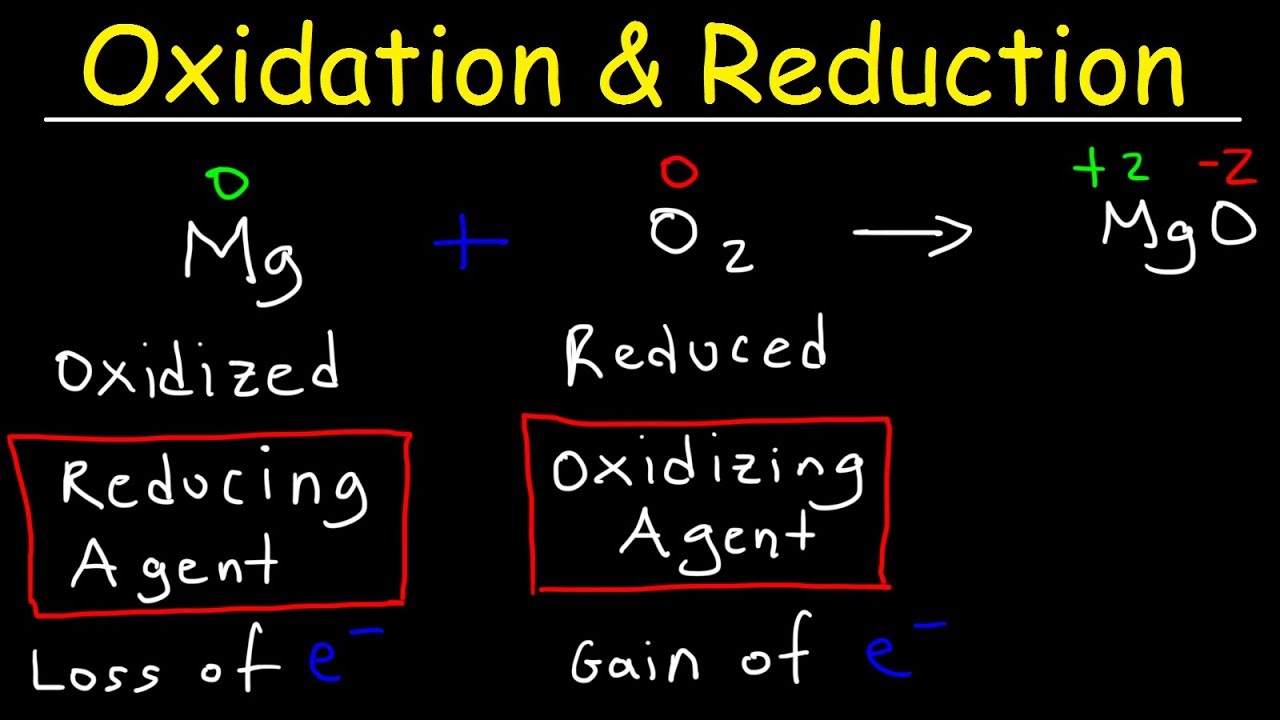
Oxidation And Reduction Redox Reactions And Electrochemistry Chemistry Khan Academy Youtube Chemistry Basics College Chemistry Redox Reactions

Pin On Halloween Science Activities

Ir Spectroscopy Functional Groups Ir Absorbance Table Http Www Chromatographytechniques Com Articles Functional Group Chemistry Help Mcat Study

Deciding Sn1 Sn2 E1 E2 3 The Solvent Master Organic Chemistry Organic Chemistry Books Medical School Preparation Organic Chemistry

Afbeelding Covalente Bindingen Overzicht Lineair Planair Tetrahedraal Etc Chemistry Chemistry Lessons Molecular Geometry

Country Report Template Middle School 2 Templates Example Templates Example Country Report Report Template Report Writing













